Man Versus Flu: The Search for a Universal Flu Vaccine
- By Keith Berman, MPH, MBA
In This Article:
Let me tell you the secret that has led me to my goal: My strength lies solely in my tenacity.
— Louis Pasteur
NO ONE CAN argue that there are not plenty of options these days for annual immunization against the flu. For the 2013-14 season, seven manufacturers — up from four a decade ago — are offering a smorgasbord of inactivated and live-attenuated and recombinant, trivalent and quadrivalent, standard and high-dose, and intramuscular and intranasal and intradermal flu vaccines.
Yet despite the panoply of choices, influenza remains a serious public health threat. The most recent 2012-13 flu season presents a sobering reminder of the limits of our current vaccine technology. While flu vaccines were 56 percent effective for all recipients, they were just 9 percent effective for persons age 65 and older. The number of flu-related pediatric deaths during last year’s flu season was the highest reported since data collection began in 2004.1 The hospitalizations avoided and lives saved by current-generation flu vaccines unquestionably justify the annual effort and cost of hunting down target strains, then manufacturing and administering around 135 million vaccine doses this season.2 But this winter, tens of millions of Americans will come down with the flu (including many, of course, who neglect to get their annual flu shot), millions of workdays will be lost, as many as 200,000 or more people may be hospitalized, and thousands will die from flu-related complications.
The obvious need remains for “a vaccine that you don’t have to give every year that works better,” said Dr. Joseph Bresee, a senior epidemiologist in Centers for Disease Control and Prevention’s (CDC’s) Influenza Division.3 “Among the two dozen vaccine-preventable diseases, including measles, mumps, polio, smallpox and hepatitis, seasonal influenza is the only one for which a new vaccine is recommended every year. A more efficient approach is long overdue,” noted Dr. Anthony Fauci, director of the National Institute of Allergy and Infectious Diseases.4
Possibly the best evidence for the feasibility of a longer-acting, broadly protective “universal” flu vaccine comes from a natural experiment — the surprisingly mild 2009 H1NI flu pandemic. Antigenically distinct from recently circulating H1N1 seasonal flu viruses, that H1N1 virus turned out to be a distant relative of the virus responsible for the devastating 1918 Spanish flu pandemic. A new analysis by scientists at the Wistar Institute has revealed that people of different ages experienced vastly different antibody response rates to the 2009 H1N1 pandemic virus, depending on whether they were exposed to a seasonal H1N1 virus many decades earlier.5 This new understanding that our immune systems can “remember” and generate neutralizing antibodies to similar-looking flu strains has bolstered enthusiasm about the prospects for a universal vaccine or vaccine cocktail.
Influenza Viruses as Escape Artists
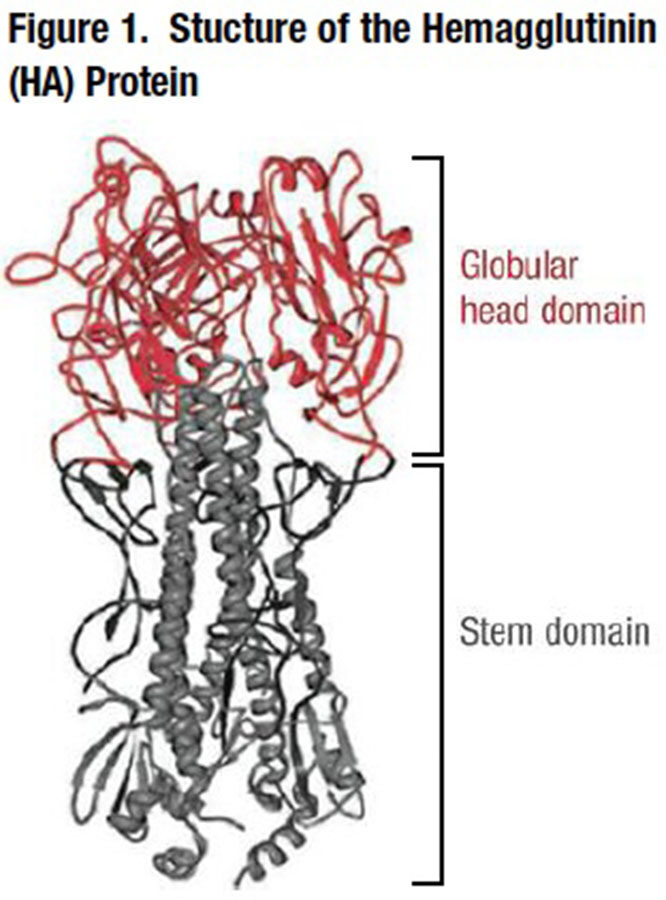
Despite their different production processes, presentations and delivery options, all licensed influenza vaccines work by inducing protective humoral immunity against antigenic targets on the globular “head” portion of hemagglutinin (HA), a surface glycoprotein that the virus uses to attach itself to respiratory tract and other host cells (Figure 1). When exposed to the circulating live flu virus, the immunized individual rapidly produces specific antibodies that inhibit virus infection by blocking HA attachment to our cells. Yet, when the next flu season rolls around, without immunization against the new epidemic strain, we are once again susceptible to contracting the illness.
Influenza viruses exploit three potent evolutionary strategies that enable them to return to infect us again and again. The error-prone replication of viral RNA strands in the human or other mammalian host cell generates spontaneous mutations that translate into changes focused in particular in the polymorphic head portion of the viral HA surface protein. Antigenic shift occurs when significant mutations create new flu viruses that are poorly recognized by large segments of the population.
As a growing portion of the flu-infected — or vaccinated — population builds effective neutralizing antibodies against the prevalent influenza virus strains, natural selection favors new variants that are capable of escaping host immunity by virtue of their altered HA protein structure. Thus, we see antigenic drift of the flu viruses that circulate over the course of each season. The greater the mismatch between the drifted virus strains and the early virulent strains against which the season’s flu vaccine was manufactured, the more the efficacy of the vaccine is diminished.
Periodic flu pandemics occur when entirely new influenza virus subtypes are created by genetic reassortment when two different viruses co-infect the host cell. Unlike most viruses that have a single RNA strand, the flu virus includes eight RNA strands to encode its genome. When all those strands intermix, a novel “reassortant” progeny virus can be created that never existed before. The H1N1 strain that caused the 2009 swine flu pandemic, for example, was a complex reassortant of avian, human and swine influenza viruses that further reassorted with a Eurasian swine flu virus.6 But as a growing share of the population was exposed to it, immune selection resulted in antigenic drift of the 2009 H1N1 virus, resulting in its transition to a seasonal flu virus.
Thus, the flu virus rebounds to create new epidemics each year by continually changing its antigenic presentation, which both necessitates the annual licensure and production of new vaccines, and leaves us highly vulnerable to the next flu pandemic. But encouraging recent findings reported by some laboratories have raised hopes that a long-dreamed-about strategy of targeting highly conserved viral proteins could yield broadly protective “universal” vaccines that confer long-term protection against both drifted and pandemic influenza A and B.
Universal Flu Vaccine Candidates
The functional premise of most development-stage universal influenza A and B vaccines is simple: Induce highly cross-reactive antibodies against highly conserved antigenic targets across different influenza virus subtypes and strains (Figure 2).
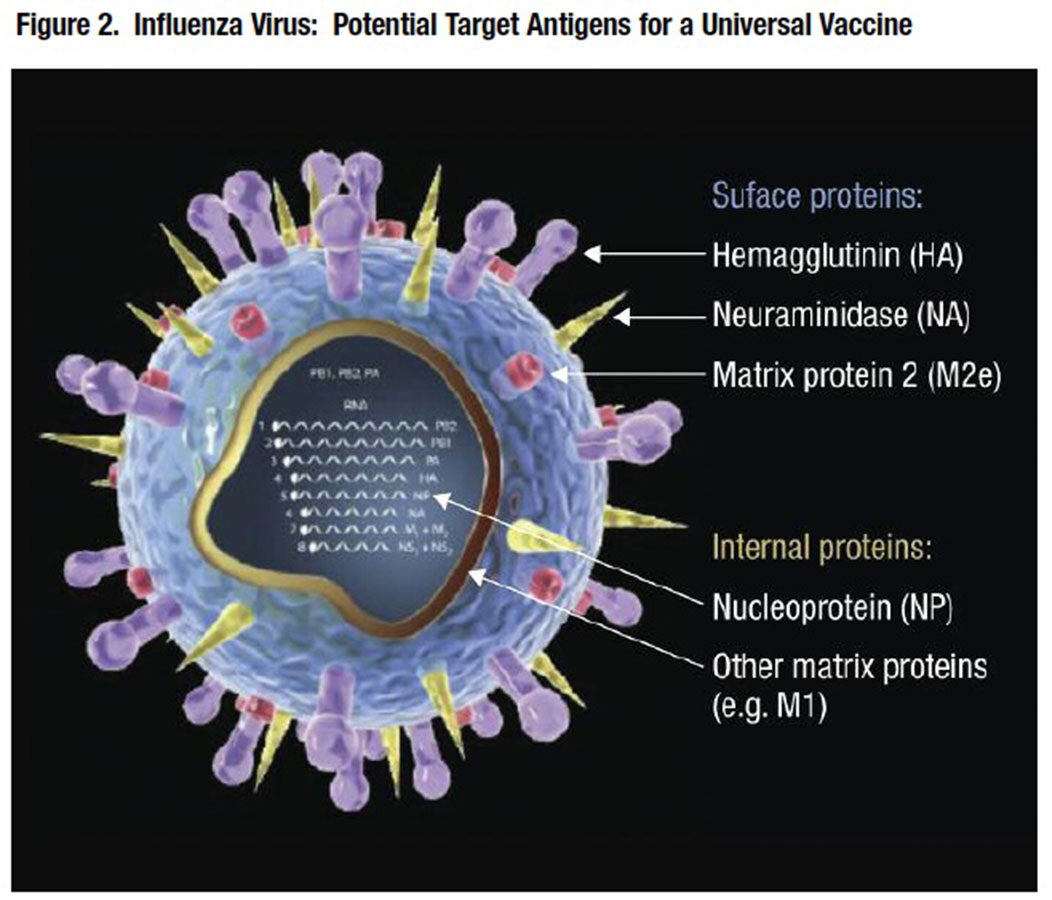
Most promising development-stage vaccines incorporate one or more of the following three viral proteins:
- Hemagglutinin A (HA) stem region. Also referred as the HA stalk, this region of the HA glycoprotein is highly conserved. One group has designed a novel vaccine that includes the stem portion of HA without the globular head; mice vaccinated with this “headless HA” were completely protected against a lethal influenza virus challenge.7 Separately, researchers at the Scripps Research Institute report isolating a monoclonal antibody (MAb) that recognizes a highly conserved epitope on the stem region of HA and neutralized influenza virus by preventing virus-host membrane fusion.8 They and others have subsequently identified other MAbs that target the HA stem region, creating an entirely separate theoretical opportunity to produce a MAb cocktail that could be used to provide passive immunity in cases of severe influenza.9
- Matrix protein 2 (M2e). This highly conserved external domain of this influenza A surface glycoprotein plays a key role in virus morphogenesis and assembly. On hopes that it can induce cross-protection against different subtypes, M2e has been selected for a number of universal influenza A vaccine candidates. To improve its immunogenicity, several groups have linked M2e to such platforms as hepatitis B virus core, rotavirus fragment10 and flagellin isolated from a salmonella species.11 Interestingly, while antibodies specific for M2e rarely occur as a natural response to infection with influenza A virus, animal and human studies have shown it is possible to induce them through vaccination.
- Nucleoprotein (NP). NP has been included together with M2e or other matrix protein in several vaccine candidates. While NP induces an antibody response, it is a vigorous CD8+ T-cell response in both mice and humans that is thought to account for evidence of protective immunity.12,13
Active vaccine development programs are scattered across university-based laboratories and start-up biopharmaceutical firms such as VaxInnate, Okairos, BiondVax, FluGen and Immune Targeting Systems. But despite universal agreement about the overwhelming need, to date, most large manufacturers of seasonal influenza vaccines have hesitated to make a major commitment to develop a universal flu vaccine.
Putting Progress in Perspective
Beginning a decade ago with the emergence of a highly pathogenic avian flu virus, worries about a potential pandemic helped spur a flurry of research activity focused on development of a universal flu vaccine. By the end of the last decade, cautious optimism was being expressed at research conferences that the first such vaccine could be available in as little as five years. This optimism has been tempered, however, by results of a number of early-stage clinical trials documenting evidence of poor cross-protection or weak immunogenicity of candidate HA- and M2e-based vaccines.14
Adding another note of caution, this year, FDA scientists described their work with a pig flu vaccine that induced highly cross-reactive antibodies against the HA stem region of a different subtype, resulting in enhanced — not attenuated — severe respiratory disease. The agency warned that universal flu vaccines that target the HA stem region “might enhance influenza disease rather than prevent it.”15 Clearly, there are more potential pitfalls ahead for those working in this complex area of vaccine research. There is a consensus that solving the puzzle of the influenza virus’ extraordinary evolutionary capacity to escape human immunity will require much more basic and applied research. Everyone agrees on the need for a universal flu vaccine. A key question going forward is whether and when government and industry will commit sufficient resources to succeed at what by any measure is a Herculean challenge.
Credit to Luke Noll, Director of Vaccine Product Sales at FFF Enterprises, for contributing helpful input for this article.
References
- Influenza activity – United States, 2012-13 season and composition of the 2013-14 influenza vaccine. MMWR 2013 Jun 14;62(23):473-9.
- U.S. Centers for Disease Control and Prevention (CDC). What you should know for the 2013-2014 flu season. Accessed 9/4/2013 at www.cdc.gov/flu/about/season/flu-season-2013-2014.htm.
- Szabo L and Weise E. USA needs more effective flu shots, experts say. USA Today (January 12, 2013). Accessed 8/22/2013 at www.usatoday.com/story/news/nation/2013/01/11/flu-strains-vaccine/1827677.
- Fauci A. Better flu vaccine on the horizon (September 3, 2013). Accessed 9/5/2013 at m.cnn.com/primary/cnnd_fullarticle?topic=newsarticle&category=cnnd_health&articleId=cnn/2013/09/03/health/better-flu-vaccinefauci&cookieFlag=COOKIE_SET.
- Li Y, Myers JL, Bostick DL, et al. Immune history shapes specificity of pandemic H1N1 influenza antibody responses. J Exp Med 2013 Jul 29;210(8):1493-500.
- Ma W, Kahn RE and Richt JA. The pig as a mixing vessel for influenza viruses: Human and veterinary implications. J Mol Genet Med 2009 Jan;3(1):158-66.
- Steel J, Lowen AC, Wang TT, et al. Influenza virus vaccine based on the conserved hemagglutinin stalk domain. Bio 2010;1:e00018-10.
- Ekiert DC, Bhabha G, Elsliger MA, et al. Antibody recognition of a highly conserved influenza virus epitope. Science 2009;324:246-51.
- Gilbert SC. Advances in the development of universal influenza vaccines. Influenza Other Respi Viruses 2013 Sep;7(5):750-8.
- Andersson AM, Håkansson KO, Jensen BA, et al. Increased immunogenicity and protective efficacy of influenza M2e fused to a tetramerizing protein. PLoS One 2012;7(10):e46395.
- Turley CB, Rupp RE, Johnson C, et al. Safety and immunogenicity of a recombinant M23-flagellin influenza vaccine (STF2.4xM2e) in healthy adults. Vaccine 2011 Jul 18;29(32):5145-52.
- Carragher DM, Kaminski DA, Moquin A, et al. A novel role for non-neutralizing antibodies against nucleoprotein in facilitating resistance to influenza virus. J Immunol 2008;181:4168-76.
- Zhou D, Wu TL, Lasaro MO et al. A universal influenza A vaccine based on adenovirus expressing matrix-2 ectodomain and nucleoprotein protects mice from lethal challenge. Mol Ther 2010 Dec;18(12):2182-9.
- Wang L, Zhang H, Compans RW, et al. Universal influenza vaccine – short review. J Immunol Clin Res 2013 Aug 7;1:1003.
- U.S. Food and Drug Administration. FDA-led study describes mechanism potentially responsible for respiratory side effect associated with a pig influenza vaccine. Accessed 9/3/2013 at www.fda.gov/BiologicsBloodVaccines/ScienceResearch/ucm366625.htm.