Emicizumab Prophylaxis Efficacious and Well-Tolerated in Infants with Severe Hemophilia A Without Inhibitors

The Phase IIIb study of emicizumab shows promise for reducing risk of spontaneous and traumatic bleeds in infants with hemophilia A.
Discovery Could Predict Immunotherapy Response in Melanoma

Researchers have discovered a rare type of immune cell that may predict how likely some patients with skin cancer will respond to immunotherapy treatment.
Researchers Identify an Immune Cell That Can Attack Cancer
Researchers at City of Hope have discovered that a type of immune cell in the human body known to be important for allergy and other immune responses can also attack cancer.
Study Uses Flu Virus to Speed Up COVID-19 Antibodies
A new vaccine may help speed up the process of making antibodies against SARS-CoV-2 by using preexisting immunity to a separate virus (the influenza virus).
Study Will Test Vaccine That Kills Cancer Cells Without Side Effects
A new cancer vaccine designed to detect and fight cancer cells without traditional cancer treatment is entering Phase III clinical trials.
Study Shows New Blood Test May Predict Alzheimer’s Risk

A recent study has found that a commercially available plasma p-tau217 (phosphorylated tau 217) immunoassay accurately identified biological Alzheimer’s disease (AD).
First Cancer Patients Receive mRNA Therapy in Clinical Trial
Imperial College Healthcare NHS Trust has enrolled the first United Kingdom (UK) patients who have received an experimental messenger RNA (mRNA) therapy — a type of immunotherapy treatment called mRNA-4359 — in its Phase I/II clinical trial.
FDA Approves New Safety Labeling for Opioid Medications

FDA approved and implemented updated warning labels for all opioid prescriptions.
Study Shows Patients Vaccinated Against Ebola Are Half as Likely to Die Than Unvaccinated Patients
A new study showed patients with confirmed Ebola virus disease were half as likely to die if they were vaccinated against the virus than if they were not.
FDA Approves First Cell Therapy to Treat Melanoma
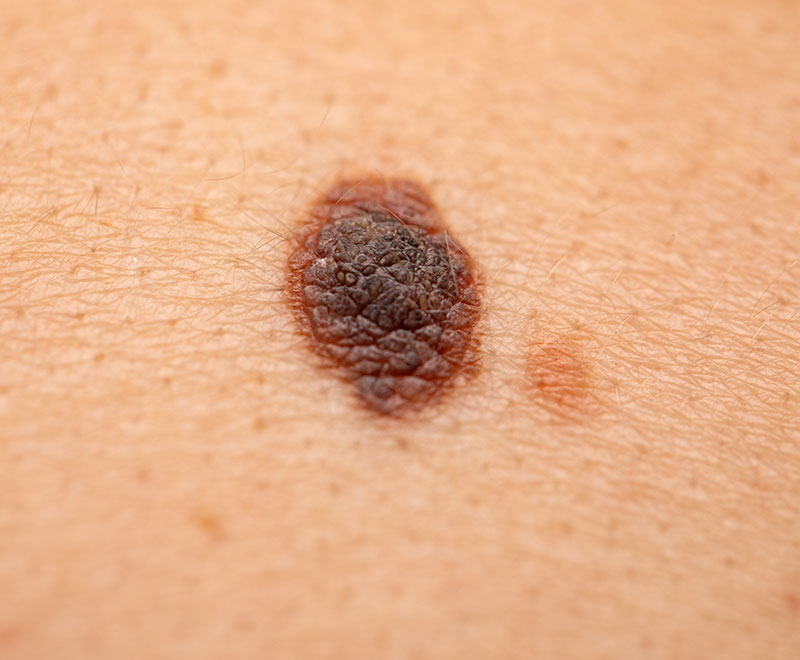
FDA approved Amtagvi, the first cellular therapy indicated for the treatment of adult patients with unresectable melanoma or metastatic melanoma that previously has been treated with other therapies.