FDA Approves Expanded Label for Jascayd for PPF

Boehringer Ingelheim’s Jascayd (nerandomilast) has been given an expanded label by the U.S. Food and Drug Administration to include use in adults with progressive pulmonary fibrosis.
Lunsumio VELO Has Been Approved for R/R FL

FDA has approved CD20xCD3 bispecific Lunsumio VELO (mosunetuzumab), as a subcutaneous formulation, for the treatment of adult patients with relapsed or refractory follicular lymphoma after two or more lines of systemic therapy.
Study Shows mRNA Vaccine Revitalizes the Immune System

Researchers have demonstrated that weakened immune systems of older individuals can be rejuvenated using mRNA technology to transform the liver in mice into a temporary source of immune regulatory factors that are naturally lost during aging.
CDC Approves Change to Nation’s Childhood Immunization Schedule

The Centers for Disease Control and Prevention has approved a change to the nation’s childhood immunization schedule to drop the long-standing practice of giving all newborns a hepatitis B vaccine within 24 hours of birth.
FDA Approves First Gene Therapy Treatment for Wiskott-Aldrich Syndrome
The U.S. Food and Drug Administration has approved Waskyra (etuvetidigene autotemcel), the first cell-based gene therapy for the treatment of Wiskott-Aldrich syndrome.
Huge Study Shows mRNA COVID Vaccines Associated with 25 Percent Lower Risk Of Death From Any Cause
In a national cohort study of 28 million individuals, researchers found no increased risk of four-year all-cause mortality in individuals aged 18 to 59 years vaccinated against COVID-19, further supporting the safety of the mRNA vaccines that are widely used worldwide.
Study Finds Tattoo Ink Affects Immune Cells and Vaccine Responses

A study that investigated the effects of tattoo ink on the body has found that the ink travels through the lymphatic system and kills immune cells, as well as alters the effects of some vaccines.
FDA Approves VOYXACT to Treat Kidney Disease
The U.S. Food and Drug Administration (FDA) has granted accelerated approval of VOYXACT (sibeprenlimab-szsi) for the reduction of proteinuria in adults with primary immunoglobulin A nephropathy (IgAN) at risk for disease progression.
Researchers Identify How to Destroy Cells That Cause Aging
Researchers at Ben-Gurion University of the Negev in Israel have discovered a way to destroy harmful “senescent” cells that accumulate with age and increase the kind of tissue damage and inflammation that slowly causes life to end.
KEYTRUDA and KEYTRUDA QLEX Approved to Treat Adults Patients with MIBC
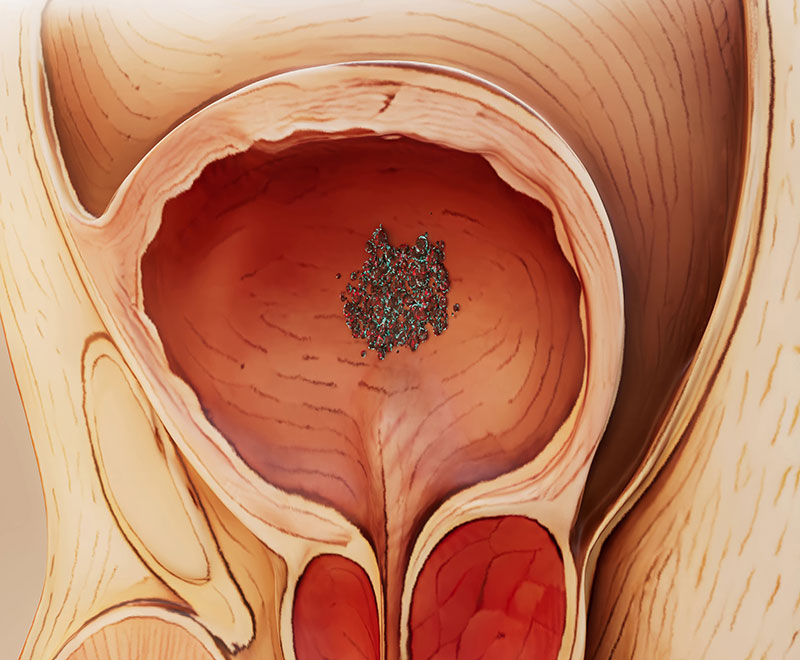
The U.S. Food and Drug Administration has approved KEYTRUDA and KEYTRUDA QLEX in combination with Padcev, as neoadjuvant treatment and then continued after cystectomy as adjuvant treatment, for the treatment of adult patients with muscle-invasive bladder cancer who are ineligible for cisplatin-based chemotherapy.