IVIG Reduces Symptom Severity for AGID

A new study indicates intravenous immun globulin (IVIG) therapy can reduce symptom severity for patients with autoimmune gastrointestinal dysmotility (AGID), also known as gastroparesis.
Addition of IVIG May Be Superior to Addition of Plasmapheresis in Corticosteroid-Resistant Optic Neuritis: Systemic Review
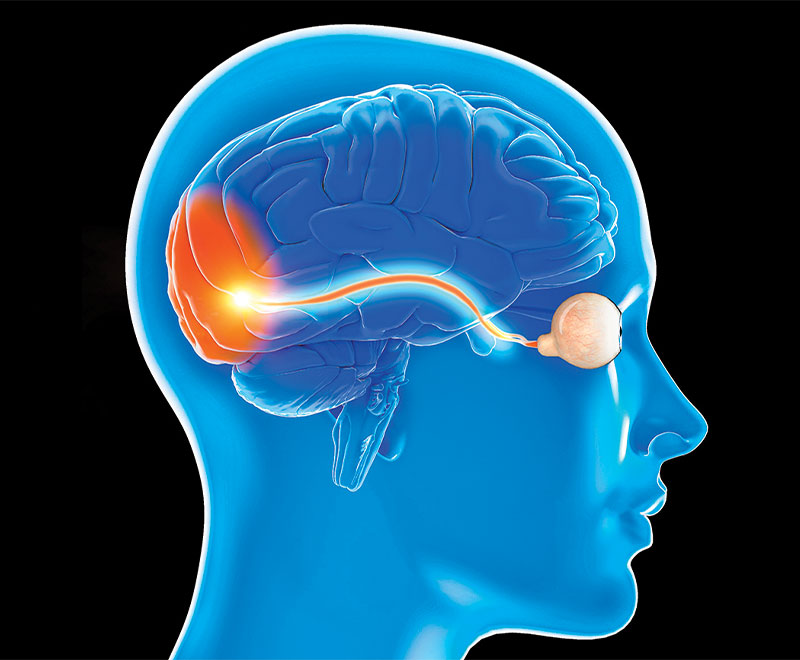
A meta-analysis and systematic review compared the efficacy of corticosteroids (CS) alone against CS combined with intravenous immune globulin (IVIG) or CS combined with plasmapheresis (PP) in patients with steroid-resistant optic neuritis.
Xembify Receives Expanded Label for PI Patients
The U.S. Food and Drug Administration (FDA) has approved an expanded label for Grifols’ XEMBIFY (20% subcutaneous immune globulin [SCIG]) to include treatment-naïve patients with primary humoral immunodeficiencies (PI).
FDA Approves Epysqli, Second Biosimilar to Soliris

Samsung Bioepis’ biologics license application for Epysqli (eculizumab-aagh) as a biosimilar to Soliris (eculizumab) has been approved by the U.S. Food and Drug Administration (FDA) to treat patients with paroxysmal nocturnal hemoglobinuria (PNH) to reduce hemolysis and atypical hemolytic uremic syndrome (aHUS) to inhibit complement-mediated thrombotic microangiopathy.
FDA Approves Yimmugo, an IVIG Product, to Treat Primary Immunodeficiencies
Biotest has received approval from the U.S. Food and Drug Administration for Yimmug to treat primary immunodeficiencies in patients 2 years and older.
The Proven Success of Immune Globulin Therapy to Treat Chronic Inflammatory Demyelinating Polyneuropathy
Numerous studies show the efficacy of both IVIG and SCIG for treating CIDP, making these the best treatment options for this rare neurological condition.
IVIG Reduces Infection Risk During Anti-BCMA Treatment for MM

Data suggests the bispecific BCMA-targeted antibody teclistamab may lead to grade 3 to 5 infections in as many as 44.8 percent of multiple myeloma patients, and hypogammaglobulinemia was noted in nearly 75 percent of patients.
Combination of IVIG and Corticosteroids Superior to Respective Monotherapies for Treatment of Relapsed ITP in Adults
The combination of intravenous immune globulin (IVIG) and corticosteroids is a more efficient and rapid treatment for relapsed immune thrombocytopenic purpura (ITP) in adults compared with the use of either therapy alone.
IVIG May Improve Neurological Symptoms in MIS-C

Recent case series findings, as well as previous studies, show that children with multisystem inflammatory syndrome (MIS-C) who present with signs of active neurological symptoms may show improvement with intravenous immune globulin (IVIG) and corticosteroids.
Study Shows Not All IVIG Products Are Effective in Treating Alzheimer’s

A recent study has found that one specific intravenous immune globulin (IVIG) product was more effective than others in counteracting the cognitive deficits, ameliorating b-amyloid (Ab) deposits and tau phosphorylation among mice with Alzheimer disease (AD)