Industry Insight
Information, Observation & Analysis
The staggering increase in autism prevalence around the world has sparked fears among parents and distorted the facts about this unexplained disorder.
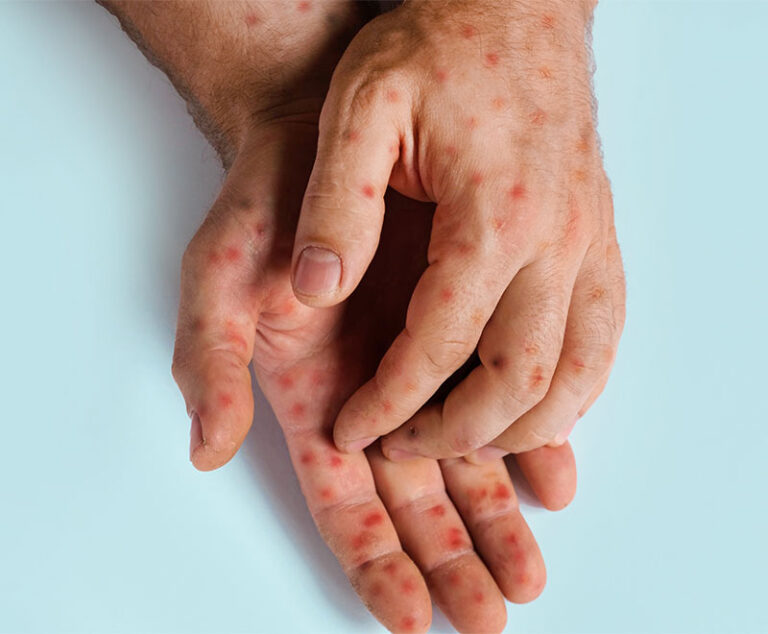
The measles resurgence is introducing a whole new generation of physicians to a once-eradicated
and potentially deadly disease.
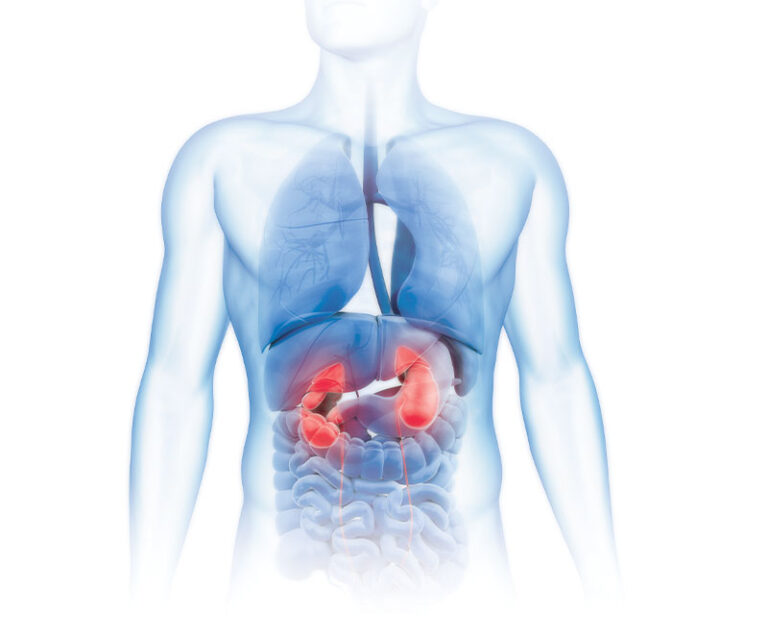
Chronic and acute kidney failure can be the result of many causes, and its prevalence is growing at historic rates, but it is no longer a death sentence.
Recent events have re-energized the ongoing debate about mandatory vaccines for children in the U.S. It remains to be seen which side of the controversy will prevail.
With more than 250 foodborne illnesses identified, it is often difficult to diagnose when a person has been infected to ensure proper treatment is prescribed.
Medicare appeals hearings from hospitals, doctors and other providers have been suspended by the Office of Medicare Hearings and Appeals (OMHA) because it has moved beneficiaries to the front of the line.
The U.S. Department of Health and Human Services (HHS) has launched the Transforming Clinical Practice Initiative, an $840 million investment over the next four years to support 150,000 clinicians.
A Kaiser Health News analysis showed that 55 percent of hospitals that were graded on the federal government's most comprehensive review of quality received bonuses, but fewer than 800 of those will actually receive the money.
The debate surrounding right-to-try laws to allow patients access to potentially life-saving drugs hinges on safety and ethical concerns.
For decades, donor blood stem cell transplantation has been the only potential cure for severe primary immunodeficiency disorders, but it has been limited by failure and serious complication risks. Now, a trifecta of scientific advances is transforming the prognosis for children once defenseless against life-threatening infections.
The president’s FY2016 budget proposal released in February would reduce net Medicare spending by $423 billion between 2016 and 2025 and is estimated to extend the solvency of the Medical Hospital Insurance Trust Fund by approximately five years.
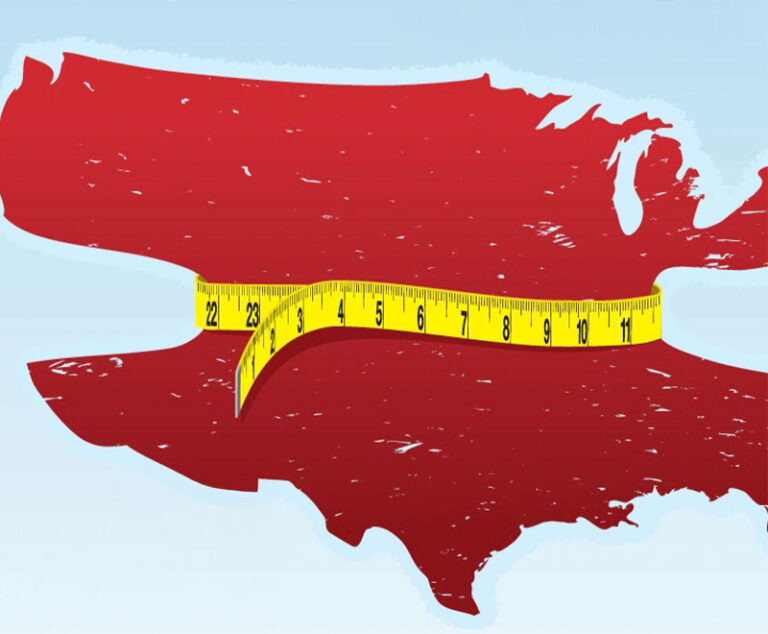
As obesity rates continue to climb, more needs to be understood to combat this costly epidemic that is now classified as a disease.