Industry News
Research, Science & Manufacturer Updates
Medicines Articles
Prophylactic treatment with an anti-inhibitor coagulant complex (FEIBA NF [Factor Eight Inhibitor Bypassing Activity], Baxter Healthcare) reduced the median annualized bleeding rate (ABR) more than three-fold as compared with on-demand treatment with the product to control acute hemorrhages, according to a Phase III study of 36 subjects with hemophilia A or B and inhibitory alloantibodies to factor VIII or factor IX.
A recent study conducted in Japan showed significant early post-administration improvements in sepsis patients who were given a single-dose administration of intravenous immune globulin (IVIG).
DiaVacs’ type 1 diabetes therapy DV-0100 has been granted orphan drug designation by the U.S. Food and Drug Administration (FDA).
A new study has found that patients who skip an antibiotic often used by hospitals to combat infection during patients’ stays and prescribed to continue that fight after discharge are more likely to return to the hospital.
Low-dose intravenous immune globulin added to a course of corticosteroids has been found to be superior to corticosteroid therapy alone in resolving toxic epidermal necrolysis and reducing associated mortality risk.
The U.S. Food and DrugAdministration has approved obinutuzumab (Gazyva, Genentech)for the treatment of patients with previously untreated chronic lymphocyticleukemia.
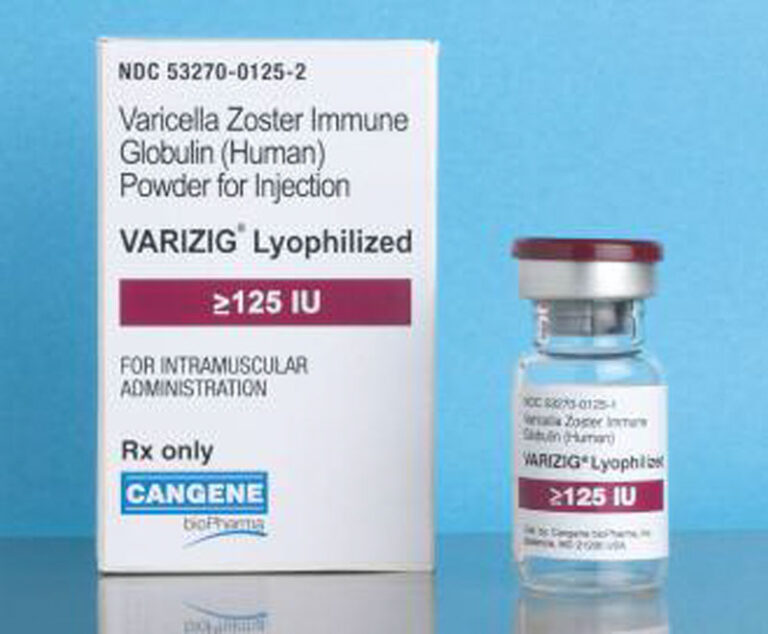
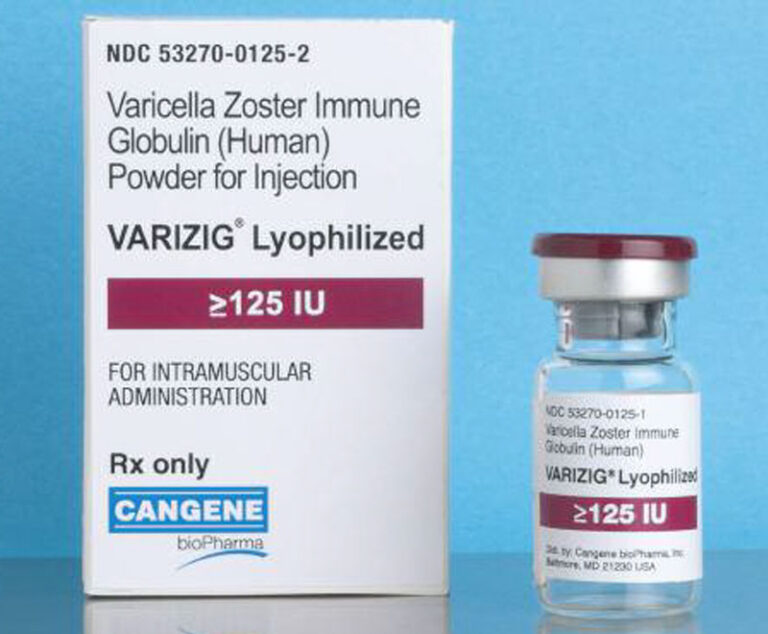
The Centers for Disease Control and Prevention has updated its recommendations for the use of VariZIG.
The U.S. Food and Drug Administration has approved a 10 g (50mL) vial size for Hizentra, immune globulin subcutaneous (human).
The U.S. Food and Drug Administration has launched the Secure Supply Chain Pilot Program (SSCPP) to enable qualified firms to expedite the importation of active pharmaceutical ingredients and finished drugs into the United States.
The U.S. Food and Drug Administration has instructed manufacturers to add information to the current boxed warning in the labeling of all intravenous immune globulin (IVIG) products, and add a similar standardized boxed warning for all subcutaneous and intramuscular IG products.
The U.S. Food and Drug Administration(FDA) has approved an expanded indication for CSL Behring’s Corifact, factor XIII (FXIII) concentrate (human) to include the perioperative management of surgical bleeding in adult and pediatric patients with congenital FXIII deficiency.
VARIZIG, a hyperimmune globulin indicated for post-exposure prophylaxis of varicella zoster virus (VZV) in high-risk patients, has been approved by the U.S.Food and Drug Administration.