Industry News
Research, Science & Manufacturer Updates
Medicines Articles
The U.S. Food and Drug Administration has approved Fluad (Seqirus), an influenza vaccine that contains the adjuvant MF59.
The U.S. Food and Drug Administration ha approved Octapharma’s NUWIQ, antihemophilic factor (recombinant), an intravenous therapy for adults and children living with hemophilia A.
The U.S. Food and Drug Administration has approved Belbuca (buprenorphine) buccal film for patients with severe chronic pain.
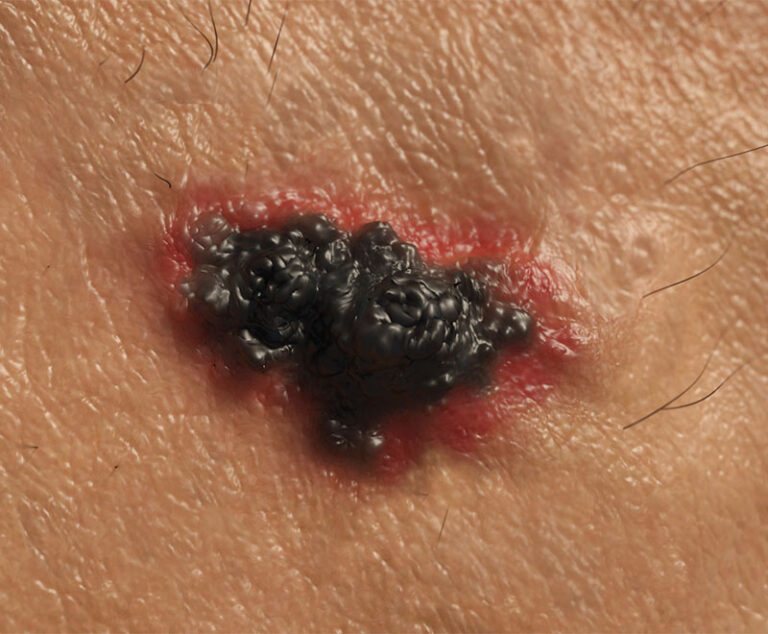
The U.S. Food and Drug Administration (FDA) approved Bristol-Myers Squibb’s Opdivo (nivolumab) in combination with Yervoy (ipilimumab) for the treatment of advanced melanoma.
The Centers for Medicare and Medicaid Services issued final rules detailing how it will pay for services provided to Medicare beneficiaries in 2016.
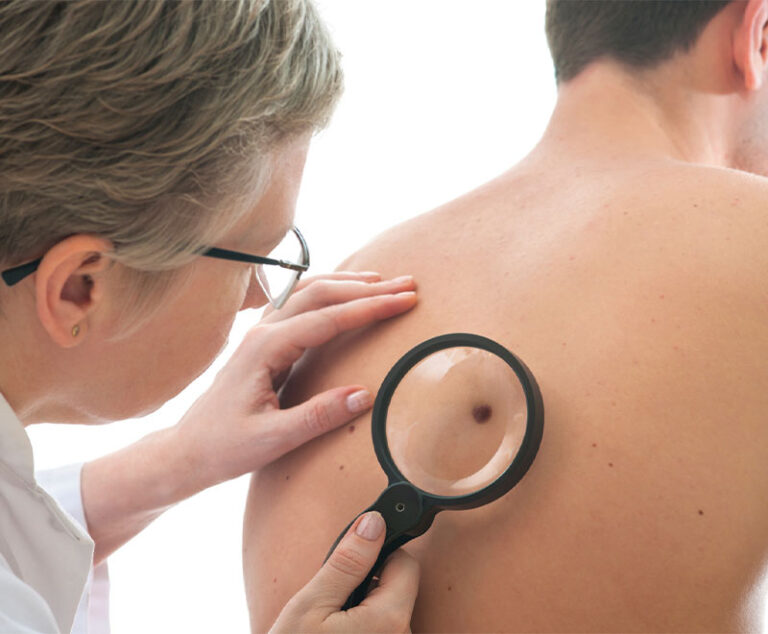
Hailed as a major advancement in the fight against skin cancer, the U.S. Food and Drug Administration (FDA) has approved a new immune-based therapy for treating metastatic melanoma.
The U.S. Food and DrugAdministration (FDA) approved BioProducts Laboratory’s Gammaplex(immune globulin intravenous [human]5% liquid) for pediatric patients 2 years of age and older who have primary immunodeficiency disease (PI).
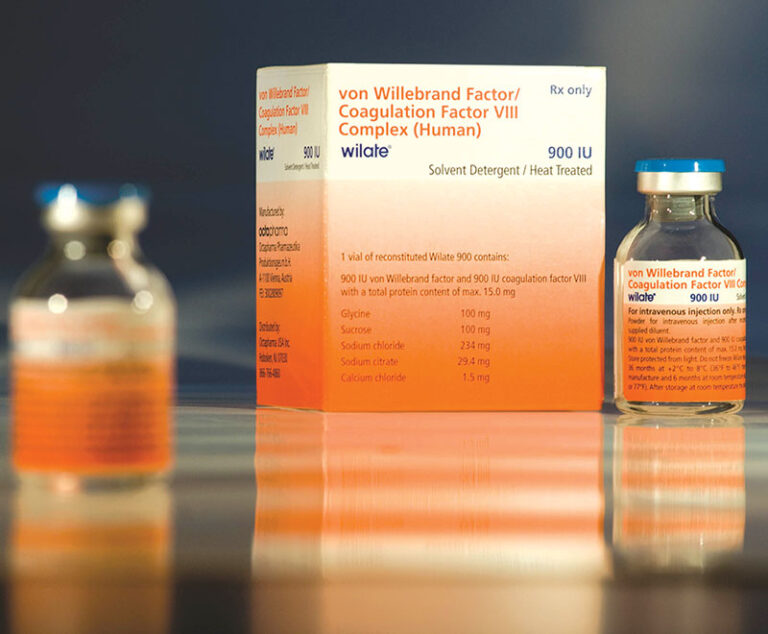
The U.S. Food and Drug Administration(FDA) has approved revised product labeling for Wilate (von Willebrand factor/coagulation factorVIII complex [human]) to include prevention of excessive bleeding during and after minor and major surgery in adult and pediatric von Willebrand disease patients.
A new report shows that stem cell transplants might soon offer multiple sclerosis (MS) patients an effective way to stave off relapses and improve their overall neurologic condition.
Scientists at the University of South Australia and colleagues from Third Military Medical University in Chongqing, China, have found that the drug Edaravone alleviates Alzheimer’s disease pathologies at multiple levels and improves learning and memory functions in mice.
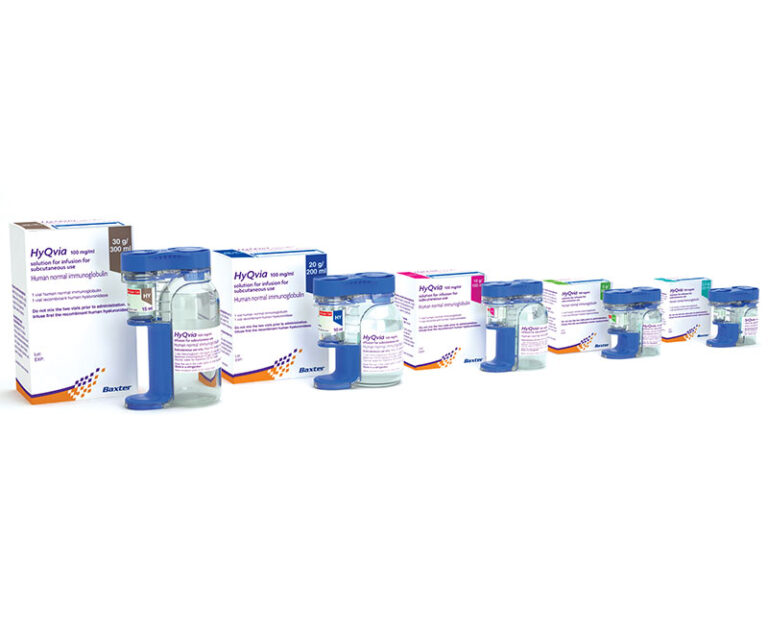
The Centers for Medicare and Medicaid Services (CMS) has expanded coverage to include in-home use of HYQVIA(immune globulin infusion 10 percent [human] with recombinant humanhyaluronidase) to treat primary immunodeficiency patients.
Pfizer (a licensee of Gliknik Inc.) has been granted orphan drug designation by the U.S. Food and Drug Administration (FDA) for its recombinant intravenous immune globulin (IVIG)-mimetic drug
GL-2045 to treat chronic inflammatory demyelinating polyneuropathy (CIDP).