Industry News
Research, Science & Manufacturer Updates
Panafina Inc.’s investigational new drug, RABI-767, has been granted fast track designation by the U.S. Food and Drug Administration (FDA) to treat patients with acute pancreatitis predicted to progress to severe disease.
Inborn errors of immunity (IEI) increase morbidity and mortality risks, particularly from respiratory tract infections. Hence, vaccination becomes pivotal for IEI patients.
Immune globulin replacement and prophylactic antibiotics are commonly used to prevent infections in patients with secondary hypogammaglobulinemia due to hematological malignancies but have never been directly compared.
Researchers at the University of Wisconsin (UW) School of Medicine and Public Health are exploring the ideal vaccine booster strategy for immunosuppressed patients to protect those at higher risk of severe illness and complications from COVID-19 infection.
A new vaccine currently in development can effectively and affordably lower levels of “bad’ cholesterol in the body, a health problem that affects almost two in five adults in the U.S.
CSL Behring’s ZEMAIRA (alpha1- proteinase inhibitor [human]) is now available in 4 gram and 5 gram vials.
The U.S. Food and Drug Administration has approved two treatments, Casgevy and Lyfgenia, representing the first cell-based gene therapies for the treatment of sickle cell disease in patients 12 years and older.
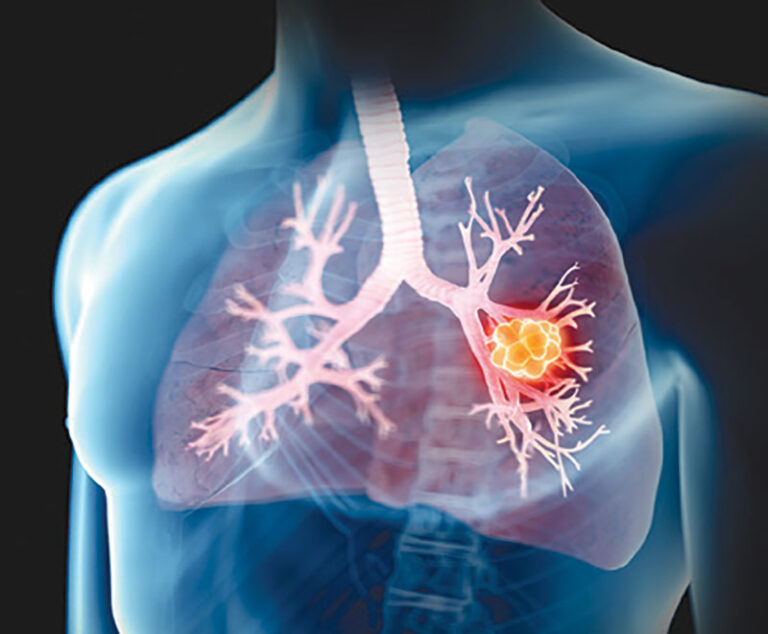
The U.S. Food and Drug Administration has approved Bristol Myers Squibb’s repotrectinib (Augtryo) for the treatment of adult patients with locally advanced or metastatic ROS1-positive non-small cell lung cancer.
The U.S. Food and Drug Administration has approved Tyenne (tocilizumab-aazg), a biosimilar referencing tocilizumab (Actemra; Genentech), to treat multiple autoimmune diseases, including rheumatoid arthritis and juvenile idiopathic arthritis.
CSL Behring is discontinuing all sizes of Hizentra vials in the U.S. by the end of September 2024.
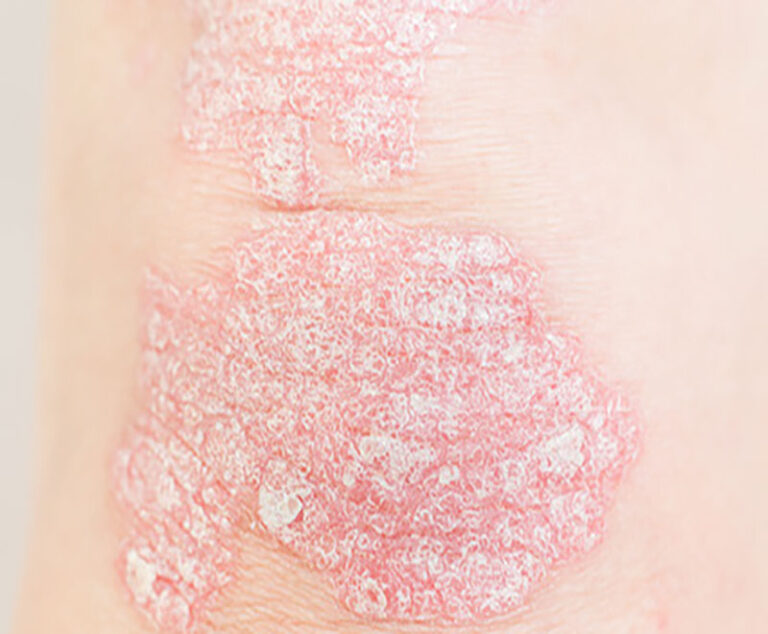
The U.S. Food and Drug Admini-stration has approved Selarsdi (ustekinumab-aekn) injection for subcutaneous use, as a biosimilar to Stelara, for the treatment of moderate to severe plaque psoriasis and for active psoriatic arthritis in adults and pediatric patients 6 years and older.
Researchers at the University of California, San Francisco, have identified a specific pattern of autoantibodies in the blood that precedes the clinical onset of multiple sclerosis.