Industry News
Research, Science & Manufacturer Updates
FDA Updates Articles
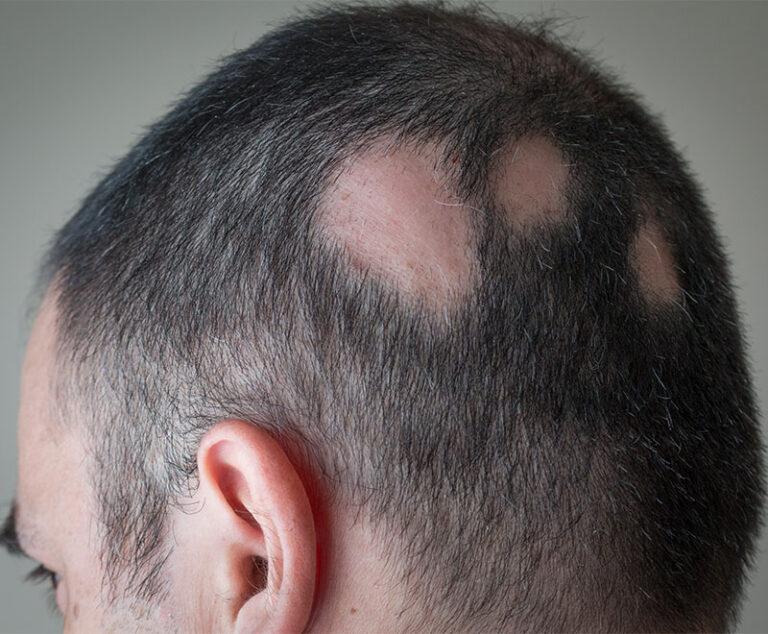
Sun Pharma’s LEQSELVI (deuruxolitinib) has been approved by the U.S. Food and Drug Administration to treat severe alopecia areata in adults.
Samsung Bioepis’ biologics license application for Epysqli (eculizumab-aagh) as a biosimilar to Soliris (eculizumab) has been approved by the U.S. Food and Drug Administration (FDA) to treat patients with paroxysmal nocturnal hemoglobinuria (PNH) to reduce hemolysis and atypical hemolytic uremic syndrome (aHUS) to inhibit complement-mediated thrombotic microangiopathy.
Panafina Inc.’s investigational new drug, RABI-767, has been granted fast track designation by the U.S. Food and Drug Administration (FDA) to treat patients with acute pancreatitis predicted to progress to severe disease.
Biotest has received approval from the U.S. Food and Drug Administration for Yimmug to treat primary immunodeficiencies in patients 2 years and older.
The U.S. Food and Drug Administration has approved a recombinant ADAMTS13 protein product (Adzynma) as a prophylactic or on-demand enzyme replacement therapy for patients with congenital thrombotic thrombocytopenic purpura.
The U.S. Food and Drug Administration has approved Eli Lilly’s tirzepatide medication, which is branded as Mounjaro for diabetes, under a new brand for weight loss as well.
UCB Pharma’s investigational agent zilucoplan, a complement C5 inhibitor, to treat patients with myasthenia gravis has been approved by the U.S. Food and Drug Administration under the market name Zilbrysq.
The U.S. Food and Drug Administration (FDA) has granted 510(k) clearance to Precision BioLogic’s CRYOcheck Factor VIII (FVIII) Deficient Plasma with von Willebrand factor (VWF), opening the pathway for the company to launch the product in the U.S.
The U.S. Food and Drug Administration (FDA) has converted Leqembi (lecanemab-irmb), indicated to treat adult patients with Alzheimer’s disease, to traditional approval following a determination that a confirmatory trial verified clinical benefit.
The U.S. Food and Drug Administration has amended the emergency use authorizations of the. Moderna and Pfizer-BioNTech COVID-19 bivalent mRNA vaccines to simplify the vaccination schedule for most individuals.
The U.S. Food and Drug Administration has approved LITFULO (ritlecitinib), a once-daily oral treatment, for individuals 12 years of age and older with severe alopecia areata.
The U.S. Food and Drug Administration has approved Roche’s Columvi, an antibody-based therapy chemically known as glofitamab.