Industry News
Research, Science & Manufacturer Updates
FDA Updates Articles
The U.S. Food and Drug Administration (FDA) has approved a new, single-dose medication to treat people 12 years and older who have had the flu for no more than 48 hours.
FDA has approved the first generic versions of Mylan’s EpiPen and EpiPen Jr.
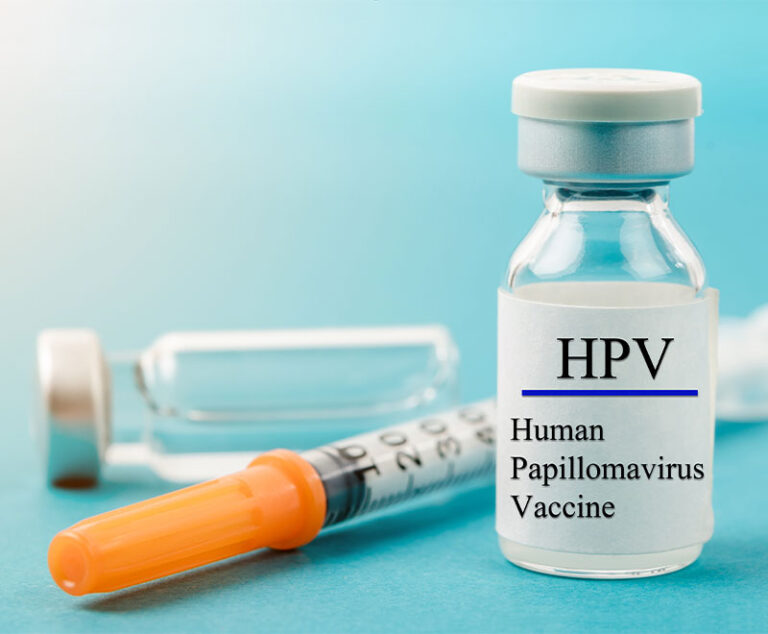
Merck’s Gardasil 9 human papillomavirus (HPV) vaccine has been approved by the U.S. Food and Drug Administration (FDA) for an expanded indication for men and women between the ages of 27 years and 45 years.
The Centers for Disease Control and Prevention's advisory committee has voted 12-2 to recommend FluMist, the nasal spray version of the influenza vaccine, be used during the 2018-19 influenza (flu) season.
The U.S. Food and Drug Administration’s (FDA) Vaccines and Related Biological Products Advisory Committee chose the Northern Hemisphere’s 2018-19 influenza (flu) vaccine strains based on the World Health Organization’s recommendations.
The U.S. Food and Drug Administration has approved Erleada (apalutamide) to treat men with prostate cancer that has not yet spread but has a quickly rising PSA level while on treatment with hormone therapy, which causes concern for cancer growth and spread.
The Food and Drug Administration has revised its recommendations for testing blood donations for the Zika virus.
The U.S. Food and Drug Administration has approved Shire’s VONVENDI, a recombinant von Willebrand factor treatment for perioperative management of bleeding in adults 18 years and older with von Willebrand disease.
The U.S. Food and Drug Administration approved Mvasi (bevacizumab-awwb), a biosimilar to Avastin (bevacizumab), the first biosimilar drug to treat cancer.
GlaxoSmithKline's Nucala (mepolizumab) has been approved by the U.S. Food and Drug Administration to treateosinophilic granulomatosis with polyangiitis (EGPA).
Abilify MyCite (aripiprazole tablets with sensor; Otsuka Pharmaceutical), the first digital ingestion tracking system, has been approved by the U.S. Food and Drug Administration.
CSL Behring noted the potential for fading print with more effect on the expiration dating on the patient tear-off portion of the vial label.