Industry News
Research, Science & Manufacturer Updates
A first-of-its-kind prostate cancer treatment that uses the body’s immune system to fight the disease has been approved by the U.S. Food and Drug Administration.
According to the Centers for Disease Control and Prevention, the rotavirus vaccine is now contraindicated for infants diagnosed with severe combined immunodeficiency.
Octapharma USA has initiated a voluntary withdrawal of all lots of Octagam (immune globulin intravenous [human] 5% liquid preparation) from the U.S. marketplace.
The Centers for Disease Control and Prevention has issued a comprehensive update on seasonal flu vaccination, which includes the new universal recommendation for everyone except infants younger than 6 months old.
A recent study shows measles-mumps-rubella-varicella (MMRV) vaccination is associated with an increased risk of fever and seizure in young children, above that already associated with measles-containing vaccines.
Updates on recent FDA approvals, appointments, acquisitions and alliances in the pharmaceutical industry.
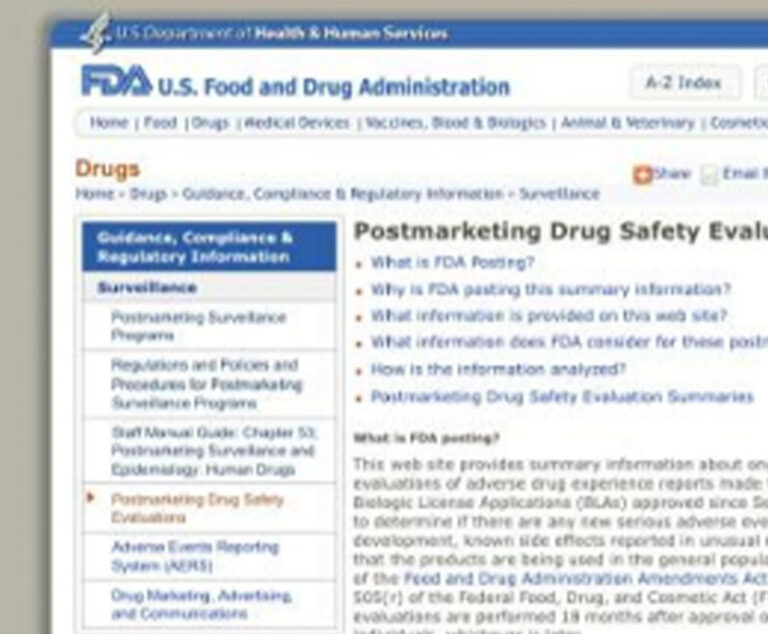
The U.S. Food and Drug Administration has launched two new websites relating to drug safety.
U.S. investigators are determining if there is evidence to support the efficacy of IVIG therapy specifically in autoimmune mucocutaneous blistering diseases (AMBDs).
The U.S. government has begun to stockpile a new version of the smallpox vaccine that is designed to close a gap that left millions vulnerable to a bioterror attack.
University of Missouri researchers are working on a vaccine to improve infants’ immune systems, which are susceptible to diseases and infections such as jaundice and E. coli, right after birth.
The Food and Drug Administration has approved two new drugs for the treatment of acute attacks of hereditary angioedema.
The recently enacted healthcare legislation contains a new incentive for biotech companies known as the Qualifying Therapeutic Discovery Project Credit (Therapeutic Credit).